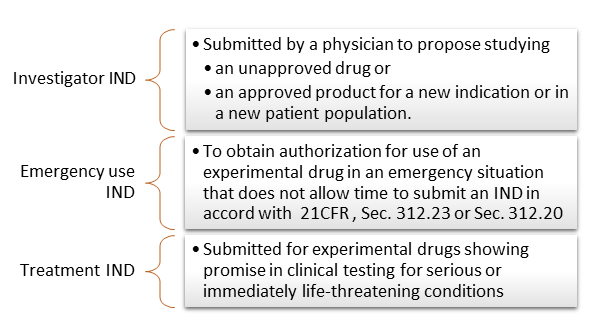
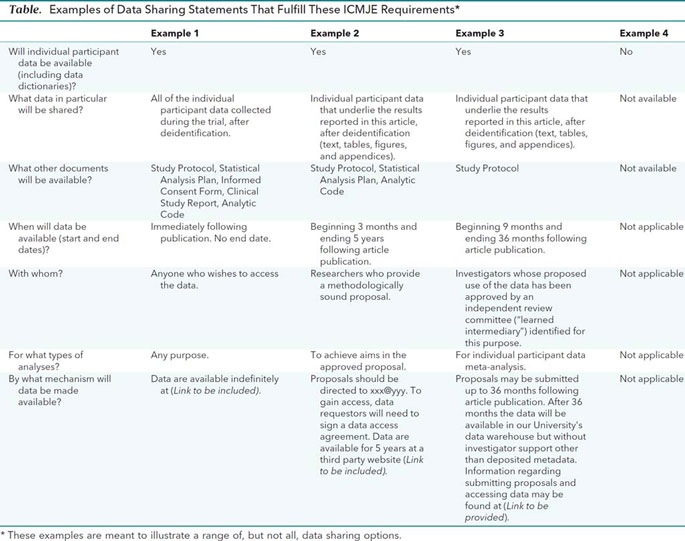
Financial ties of principal investigators and randomized controlled trial outcomes: cross sectional study | The BMJ

OCR Industry Sponsored Study Guide | Office of Clinical Research | Perelman School of Medicine at the University of Pennsylvania
Fillable Online CLINICAL INVESTIGATOR FINANCIAL CERTIFICATION. AIDS Clinical Trials Group Network IND Study Financial Disclosure Form Template Fax Email Print - pdfFiller

What are the key areas a clinical trial investigator needs to consider while reviewing the trial agreement? | Clinical trials, Research skills, Clinic