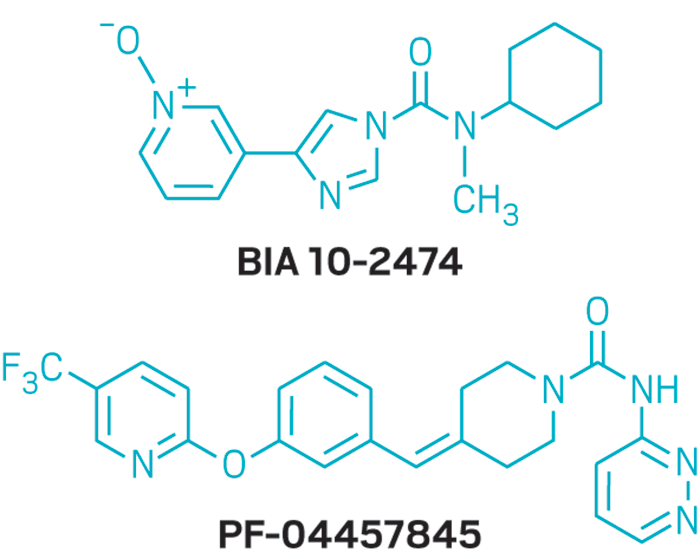
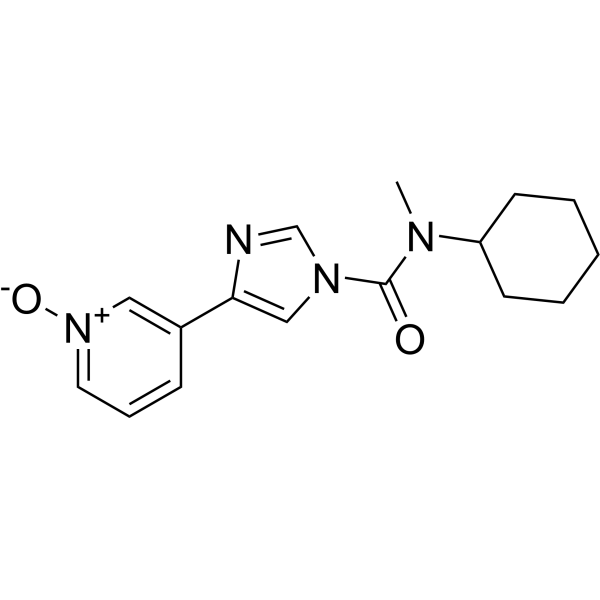
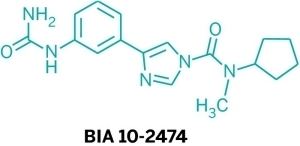
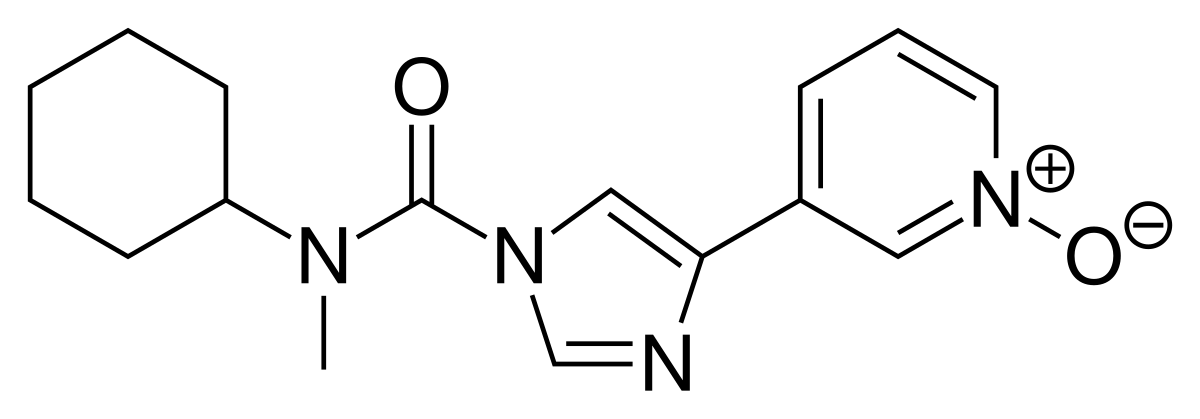
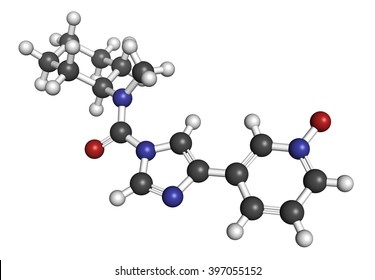
BIA 10-2474 experimental drug molecule, illustration - Stock Image - F028/2190 - Science Photo Library
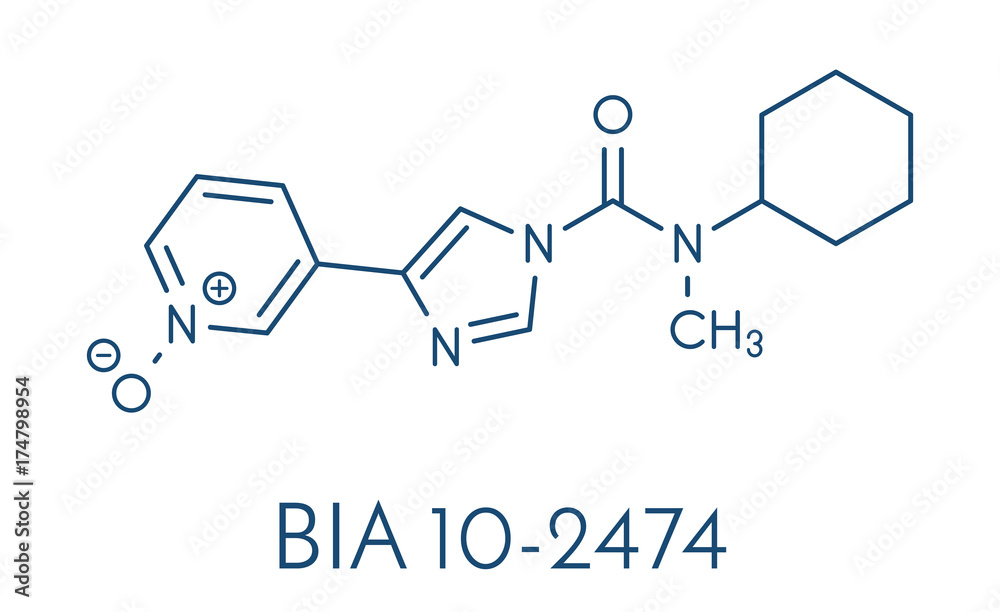
BIA 10-2474 experimental drug molecule. Fatty acid amide hydrolase (FAAH) inhibitor that caused severe adverse events in a clinical trial in France in 2016. Skeletal formula. Stock Vector | Adobe Stock

BIA 10-2474 experimental drug molecule. Fatty acid amide hydrolase (FAAH) inhibitor that caused severe adverse events in a clinical trial in France in Stock Vector Image & Art - Alamy

PDF) What failed BIA 10-2474 Phase I clinical trial? Global speculations and recommendations for future Phase I trials
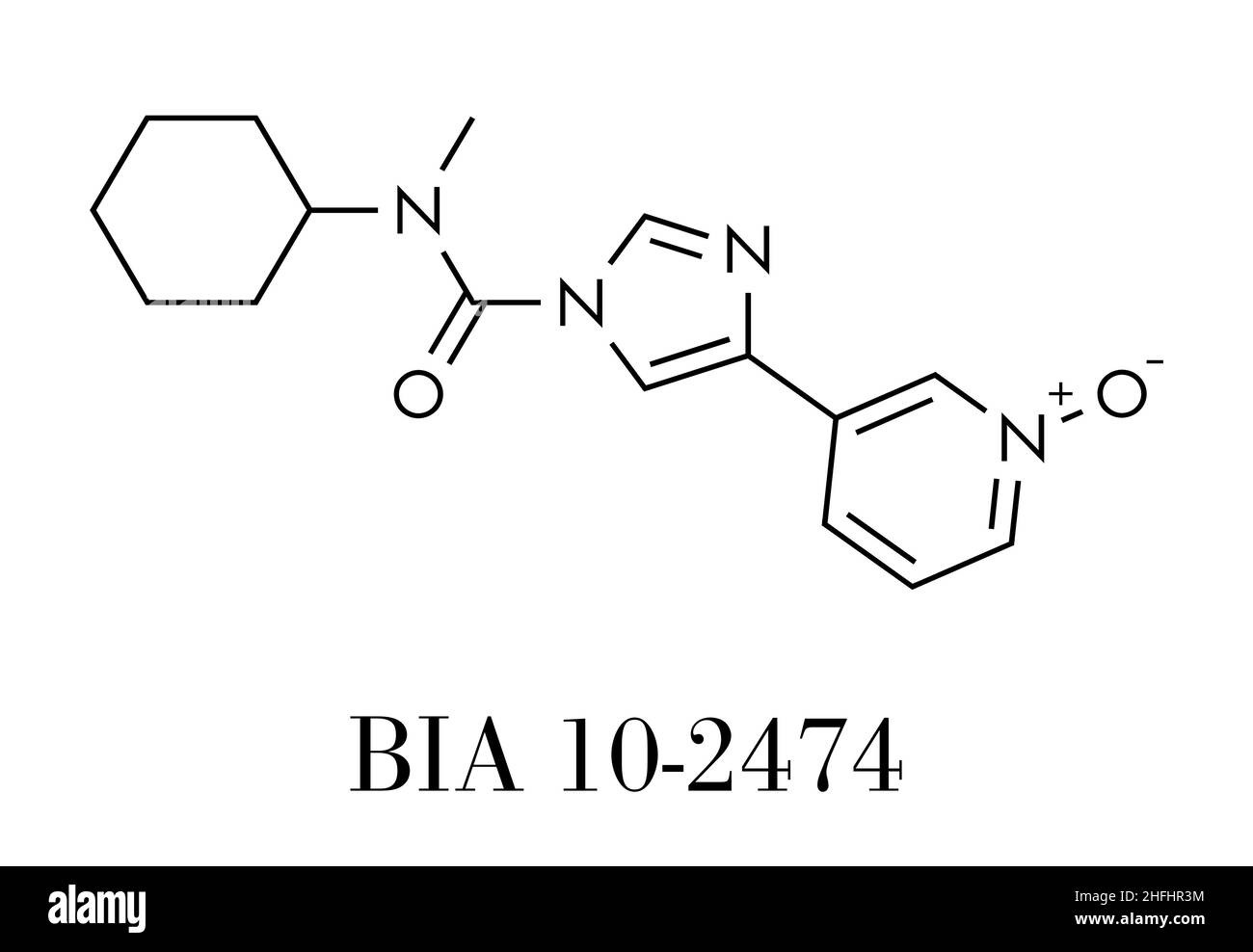
BIA 10-2474 experimental drug molecule. Fatty acid amide hydrolase (FAAH) inhibitor that caused severe adverse events in a clinical trial in France in Stock Vector Image & Art - Alamy

What failed BIA 10-2474 Phase I clinical trial? Global speculations and recommendations for future Phase I trials. - Abstract - Europe PMC

BIA 10-2474 Experimental Drug Molecule. Fatty Acid Amide Hydrolase (FAAH) Inhibitor that Caused Severe Adverse Events in a Stock Illustration - Illustration of carbon, hydrolase: 187049027
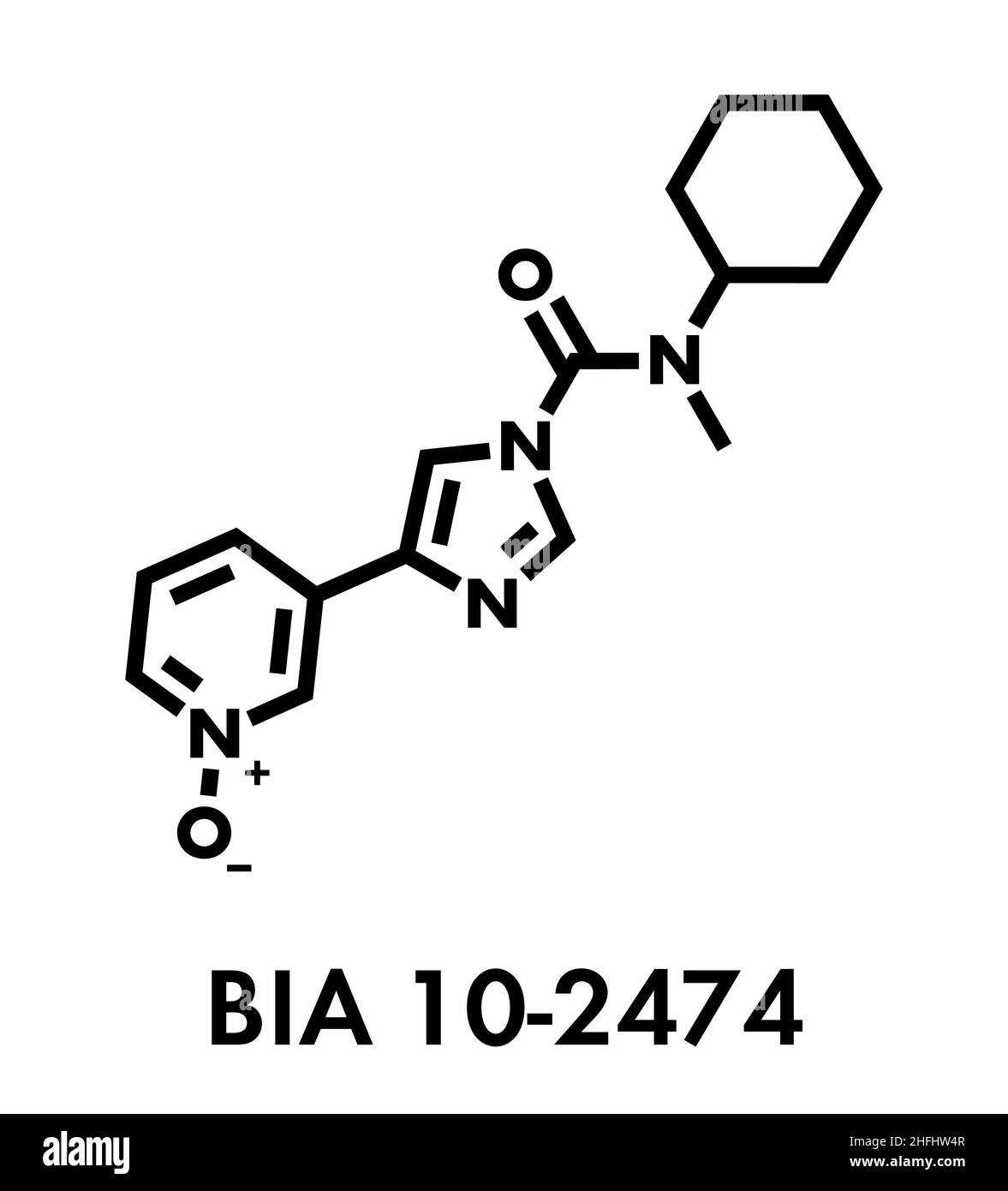
BIA 10-2474 experimental drug molecule. Fatty acid amide hydrolase (FAAH) inhibitor that caused severe adverse events in a clinical trial in France in Stock Vector Image & Art - Alamy

Man who died in French drug trial had 'unprecedented' reaction, say experts | Medical research | The Guardian